The Perfect Specimen: A Scientist's Guide to Acquiring Good Images
- Acquiring Images from Standard (Compound) Microscopes
- Laser Scanning Confocal Systems
- Flatbed Scanners
- Imaging on Stereo Microscopes
- Environmental Imaging
- Images from PowerPoint and Other Applications
VISUAL DATA INTENDED for any output contains as much detail as possible within the limits of the acquisition device and the kind of energy used (light, electrons, x-ray, etc.). To achieve the resolving of details, the specimen needs to be prepared to optimize the quality of the images, and the medium, if any, between the specimen and the imaging device needs to be appropriate for optimal imaging. As far as controls for the imaging device, the following conditions are met:
- Apertures are set (when appropriate) to optimize resolution of detail.
- The energy source is focused, if possible, so that all parts of the specimen are struck at equal intensities.
- Correct procedures are followed to minimize damage to the specimen and to create conditions for matching what is seen by eye to the visual data through attenuation of the energy source and filters.
- Methods are used to improve signal to noise.
- Dark to bright specimen features are kept within the dynamic range of the imaging device; otherwise, details are lost in areas that are too bright or too dark.
When visual data is derived from specific software applications (versus imaging devices), details are preserved under the conditions that pixel resolutions are either maintained (when images are transferred from one application to another), or dense enough to avoid seeing discrete pixels (when lower resolution images and graphics are destined for outputs).
Ways to meet imaging conditions in the preceding list using five different devices are covered in this chapter. They include compound microscopes, confocal systems, flatbed scanners, stereo microscopes, and SLR (Single Lens Reflex) cameras. For meeting pixel resolution conditions when images are derived from various digital sources, specific software applications are described, including PowerPoint).
Acquiring Images from Standard (Compound) Microscopes
While various software acquisition programs exist for each brand of camera, the hope is to find certain software components to aid in achieving three end results:
- Accurate representations of a specimen in terms of color rendition and tonal range, and optimal rendition of contrasting features (contrast)
- Evenly illuminated specimens (unlikely on microscopes)
- Images of specimens devoid of electronic noise (visible noise is more likely with dim specimens)
Accurate Representations
Optimal representation of specimens is achieved by setting up the microscope correctly and by adjusting camera settings. Specimens prepared for brightfield imaging (darker specimens on a bright background) require specific set up procedures for focusing the illumination beam (Koehler illumination). Darkfield imaging (brighter specimen on a darker background), when it is of fluorescently labeled specimens, may also require focusing of the illumination beam. Setup of the microscope for darkfield and other microscopy techniques are outside the scope of this book, but the information can be found at http://micro.magnet.fsu.edu.
Adjustment of camera settings include white balancing for brightfield, color images; determination of method for determining exposure (automatic or manual); adjusting exposure and possibly attenuating the light level so that the tonal range of the image is within the dynamic range of the camera; choosing appropriate camera settings depending on the sample; and acquiring the image to evaluate. Filters may be put in the illumination path to improve contrast within the specimen.
Even Illumination
Especially with lower magnifications, uneven illumination across the field of view is likely to be seen when evaluating the image. Illumination is evened out by applying a flatfield correction through the use of image acquisition software, if that component is available. When software components for achieving flatfield correction cannot be found in image acquisition programs, uneven illumination can be corrected later in post-processing. These methods are described in the "Flatfield Correction" section.
Noise Reduction
The presence of noise can be ameliorated or removed by using components found in acquisition software. They include background subtraction for removing hot pixels and frame averaging. But when these components don't exist, additional images can be taken to correct for noise and uneven illumination in post-processing. Corrections for uneven illumination can also be made in post-processing without additional images, but they are likely to result in a loss of image information to varying degrees.
The step-by-step procedure for acquiring images on a microscope depends on the mode of the resultant image (grayscale or color) and the technique used for illuminating the specimen (brightfield or dark-field). A flowchart that includes relevant steps is shown in Table 4.1.
Table 4.1. Microscope Imaging
|
Darkfield, Fluorescence |
Brightfield, Color |
Brightfield, Grayscale |
|
Allow equipment to warm up |
Allow equipment to warm up |
Allow equipment to warm up |
|
Set Exposure/Gain and Contrast |
Set Koehler illumination |
Set Koehler illumination |
|
Bin for more sensitivity, if necessary |
Set light source to 3200K |
Use filters, if necessary |
|
Frame Average, if noisy |
Set Exposure/Gain and Contrast |
Set Exposure/Gain and Contrast |
|
Flatfield correct |
White balance |
Flatfield correct |
|
Background subtract |
Flatfield correct |
Background subtract |
|
Background subtract |
Setting Up the Microscope
For color images, brightfield, use the following settings:
-
Brightfield, color: Set color temperature. The color temperature of the light on the microscope is best set to 3200 degrees Kelvin for consistency. It provides a discrete color temperature for the detector at which parameters for interpretation of color are well defined along a common standard. Often, markings exist for setting this color temperature. When the markings don't exist, it is likely that turning the power all the way up will create a color temperature of 3200K.
However, the light may be too bright at this setting. Look for a means to place neutral density filters in the light path. These filters block visible wavelengths equally (thus, the word "neutral") at gradients (densities) along a logarithmic scale.
- Brightfield: Set Koehler illumination. The light passing through the specimen needs to be collimated to varying degrees, depending on the lens (objective) that is used. Go through methods for setting up Koehler illumination. This method is described in detail at http://micro.magnet.fsu.edu.
Capturing Images on Camera or in Acquisition Software
The first decision that has to be made (or is instituted by the workgroup already) concerns the method for acquiring images: whether to use camera software/controls for automatic or manual exposure settings.
Automatic Exposure
Automatic determination of exposure can be used for all images except those destined for optical density/intensity (OD/I) measurements.
When this feature is chosen, cameras must read (meter) brightness or darkness levels from the specimen to determine the length of time the detector requires to collect photons to fill the dynamic range. The camera meters in several ways.
Darkest/lightest feature on specimen to avoid clipping. For many scientific cameras, the brightest or darkest value in the specimen is used to determine exposure. The exposure is determined based on having enough photons to fill detectors for values above the bottom limit for the darkest value, and not too many photons to overfill detectors at the top limit.
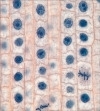
The net result of this method for metering is that bright or dark artifacts can skew the exposure so that images are overall darker or lighter, respectively. In Figure 4.1 the image on the left demonstrates overall lightening as a result of the meter exposing to keep dark spots (artifacts) within the dynamic range—shown most dramatically in the loss of black borders around the features and gain in overall contrast. Exposure is correct on the image on the right where no dark spots confuse the meter reading.

Figure 4.1 An image showing mistaken exposure reading as a result of artifacts (left); a correct reading is on the right.
When the exposure is misread, the following approaches can be taken:
- Find a different location on the specimen.
- Find a way to override the Automatic Setting by adjusting exposure time. Figure 4.2 shows an Adjustment Factor to decrease overall exposure, thus compensating for the misreading of exposure.
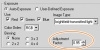
Figure 4.2 Exposure dialog box showing a 5% reduction in the Adjustment Factor.
- Use the Manual feature.
Weighted average or spot metering. Consumer cameras often provide different options based on an average. These can be set (often the default setting) to find an average of brightness and darkness values to determine exposure, generally metering from the lower part of the specimen, because this method is often most reliable for landscapes with a bright sky. Or varying patterns for metering down to a narrow, circular pattern (spot metering) can be chosen and used.
Depending on the specimen, these options can be effective, but significant gray or color values can be clipped!
Manual Exposure: Setting Dynamic Range Limits
Manual determination of exposure is used when subsequently measuring OD/I values. It is also used when correcting for noise and uneven illumination. Certain workgroups may also use manual features for consistent imaging of specimens. In a workgroup scenario, the exposure is set once and then stays at that setting over time, or for each imaging session.
Automatic exposure or auto gain features must be turned off to use manual settings; alternatively, a manual setting is chosen. When determining exposure, any one of three situations—or a combination—arises:
- Exposure is set interactively when the image is live on the screen.
- Specimen exposures are calculated through a software feature as a starting point, after which exposures may have to be iteratively determined.
- Exposures are determined by trial and error only.
The brightest and darkest significant values in the image can be measured to determine whether they remain within the dynamic range of the detector. Most acquisition software includes an interactive way to place a cursor over the brightest/darkest area and then to read grayscale or color values. If a means is not available, these images can be saved, and then opened in Photoshop to obtain measurements. A means may also be available for placing a color overlay on the image to show specimen areas that are clipped while the image is live.
For imaging a set of specimens in which OD/I will be measured, the manual setting is determined by finding the brightest labeled specimen (darkfield) or the darkest labeling (brightfield) from a set of specimens. That setting then remains the same for all subsequent specimens with adjustments only for fluctuations in the light source or when new specimens from a different experimental set exceed the dynamic range of the detector (see Chapter 3, "Guidelines for Specific Types of Images").
Darkest significant values are set above 0. Brightest significant values depend on the bit depth of the camera, as shown in Table 4.2.
Table 4.2. Grayscale/Color Limits for Significant Bright Areas on Specimen
|
8-Bit |
12-Bit |
16-Bit |
|
254 |
4094 |
65,534 |
Color, Brightfield: White Balance
To correctly compensate for the color temperature of the light source, a calibration step is done. The calibration is to the color white. Once this color is correctly interpreted and displayed, all other colors should then be displayed correctly. This step can be done automatically or manually. The manual method is preferred, because it is more precise.
Automatic. Some cameras can be set to auto white balance, which appears in some acquisition software as AWB. When this setting is used, the camera attempts to read from the specimen to interpret colors, or the color balance is obtained via a predetermined setting when the white balance feature is set to incandescent light (3200K light).
If auto white balance does not correctly interpret colors, use manual white balance. Note that cameras may auto white balance after using a mouse to outline a region of your specimen that is white, as in Figure 4.3 where the image on the left contains greenish background values: When corrected by outlining the white balancing area with a mouse (shown by a rectangular outline) and then clicking the AWB button, the image on the right is the result.
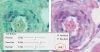
Figure 4.3 Images showing the result (right) when using auto white balance.
Manual. Other cameras white balance after the specimen is removed from the light path. In that instance, the light source is used as the white reference.
Camera Settings
Additional camera settings may or may not be available in acquisition software for cameras (Figure 4.4). A list of settings that may be available follows:
- Gain. Gain might be available in image acquisition software instead of, or in addition to, exposure. Increases in gain result in images that are brighter overall. Because gain is an electronic amplification of signal, noise levels accompany higher gain settings. Determine the highest gain setting possible without the introduction of noise and stay beneath that setting if possible. Use exposure, gain, or both to increase brightness of a specimen with the least possible addition of noise when the specimen is dim.
- Contrast. Contrast works like a Brightness/Contrast setting in many image processing programs by increasing the brighter pixels (making them brighter) above the midpoint and decreasing darker pixels (making them darker) below the midpoint. Alternatively, the acquisition program can increase contrast by incrementally darkening from the darkest black to the brightest white in the image with the greatest change occurring in the darker regions and little change to the brightest regions. This can be done with greater precision in Photoshop.
- Black. This setting is an electronic function that is used as a means to set the darkest black value. This option is set so that no value in the image reads zero, because that would clip the darkest values. Set the darkest values so that they are slightly above zero.
- Exposure (or Integration; sometimes named Brightness). The brightest values are changed by adjusting Exposure. This option attenuates the amount of time the detector is exposed to light from the specimen. Attenuate Exposure while measuring the brightest significant values so that values do not exceed the dynamic range of the instrument (clip).
- Readout Speed. This setting adapts the camera to the capabilities of the computer to show live images. A slower readout allows the computer more time to display new images (refresh) when viewing images live. Higher speeds may increase the noise level of the image, depending on the capabilities of the electronics in the camera, so lower speeds are advised.
- Image Depth. This setting is the same as bit depth and should be set to higher values.
- Preview. This option allows for a smaller display of the image in the event that the size of the image overfills screen resolutions. For example, if the screen resolution is set at 1024 x 768 and the live image is at 1380 x 1035, the image will be larger in dimension than the screen, and the outer parts of the image will not be visible.
- Bin. To increase sensitivity, discrete fractions of the image can be specified with a resulting increase in the number of sensors used for each pixel. For example, if a Bin of 2 is used, each resultant pixel would then be the sum of the photons from four sensors (two horizontal, two vertical). The image size would then be one fourth the total number of pixels (half horizontally and vertically), leading to less pixel resolution but a consequent increase in the sensitivity to light from the specimen. Bin the image when specimens are dim and the image is noisy.
- Gamma. The Gamma setting is typically kept at 1 to maintain a linear relationship of tonal values, which is absolutely required for OD/I destined images. When the linear relationship is not required or is not as important as being able to see visual information in darker or brighter areas, gamma can be altered (for publication, the Gamma setting is reported). A gamma adjustment alters midtones while maintaining detail in brighter and darker areas. Gamma is a reference to the Greek symbol used in a math formula in which the numeric value chosen becomes a power function. The gamma formula is applied after tonal values have been assigned to the pixels (after the electronics have read out the brightness values for each pixel), which can be done in post-processing with more precision in Photoshop.
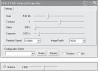
Figure 4.4 A settings dialog box in acquisition software showing camera settings.
Acquire the Image
Once settings are determined, acquire the image. Once the image is acquired, it can be evaluated visually and then other steps can be taken to improve the quality of the image. Chief among these steps is a correction of uneven illumination (flatfield correction) across the length and width of the image, which is difficult to see by eye. This correction is recommended for all images from microscopes.
Images and additional steps that can be taken to improve image quality follow (Figure 4.5):
- Accurate image. If the image appears to be an accurate representation of the specimen, consider producing a flatfield image, which is crucial for quantification and image stitching. See the "Flatfield Correction" section to produce evenly illuminated specimen.
- Dim and noisy image. If the specimen is dim and the resultant image contains visible noise levels, see the "Noise Reduction" section.
- Binning. If the specimen is so dim that gain and exposure create unacceptable noise levels, consider binning.
- Uneven illumination. If the specimen is unevenly illuminated, see the "Flatfield Correction" section.
- OD/I image. If the image is measured for OD/I, remove or reduce noise and flatfield correct.
- Incorrect colors. If the specimen is in color and colors are not correct, white balance again. If coloring changes across the width of the image, the illumination bulb may not be centered (ask your microscope representative for assistance).
- Contrast. If stains do not contrast with background information or if the specimen is lacking in contrast (or the contrast is too high so that details are obscured in darker or brighter areas), adjust gamma or contrast in the camera acquisition software or in Photo-shop. If the image is in Grayscale mode, consider using filters. See the "Grayscale: Filters" section.

Figure 4.5 Examples of problematic images and techniques that can be used to fix them.
Flatfield Correction
Light sources often produce uneven illumination across the field of view. This is rarely observed by eye and is not often seen in the image. After the unevenness is corrected—variously called flatfield, shading, or blank field correction—the appearance of the image is not only improved but made ready for quantification and image stitching (also called montaging). To correct for uneven illumination, the flatfield image is divided into the specimen image, and then multiplied by a constant. Often, this is done after subtracting a background image.
Flatfield correct in image acquisition software. Check for the ability to specify a flatfield image for subsequent use in correcting uneven illumination in acquisition software (Figure 4.6). If flatfield correction can be done in software, for brightfield specimens, acquire the flatfield image by moving to an empty area of the slide sans debris. Defocus slightly, if necessary.
Figure 4.6 Examples of Flatfield capture dialog boxes in image acquisition software.
For fluorescently labeled specimens, remove the microscope slide, and then insert a fluorescent reference slide either consistent with the wavelength range of the labels for the specimen (when neutral density filters can be placed in the light path because the emissions light is often too bright for the detector) or inconsistent so that only bleed-through emission light is collected (not as bright). Focus below the surface of the slide so that detritus and scratches on the surface are defocused.
Saving flatfield image for subsequent correction. If the camera software does not have flatfield correction, save and date the flatfield image. Make sure that no pixel value exceeds the dynamic range of the instrument (e.g., values at 254 for an 8-bit camera system): Values can often be found in image acquisition software. Flatfield images can be used for correction of uneven illumination later in Photoshop (see "Uneven Illumination Correction" in Chapter 6).
Noise Reduction
Several sources of noise are present in images, but two more common sources are mentioned here. Random noise is due to fluctuations of a finite number of photons and electrons from one moment to the next when that number is low, such as when specimens are dim. Fixed pattern noise appears when certain pixels become bright. These pixels are called "hot pixels" and are most apparent at long exposure times.
Darkfield, frame averaging to correct for random noise. To correct for random noise, more than one image is taken and they are averaged together. If the frame averaging option is available in camera software, the number of images (frames) are specified and the images are automatically taken and then averaged (Figure 4.7). If not, several images must be taken for subsequent averaging (see "Frame Averaging to Reduce Noise" in Chapter 6). Brightfield specimens are generally well illuminated, and images are more likely to be devoid of random noise. Noise reduction is more often necessary for fluorescently labeled specimens.
Figure 4.7 An example of a Multi-Frame dialog box that shows averaging of four frames to reduce random noise.
Fixed pattern noise: background subtraction. To remove hot pixels, an image is taken in ambient light with the light source blocked. The image is taken at the same setting as the time required for imaging the specimen, which was determined earlier. It may be difficult if not impossible to see hot pixels, but when the dark image is brightened, these pixels become visible. The dark image is subtracted from the specimen image. The common reference to this procedure is "background subtraction" (Figure 4.8).
Figure 4.8 Examples of dialog boxes for acquiring a background subtraction image and for naming background and flatfield images.
If the ability to background subtract is not available in image acquisition software, save and date the dark image for both darkfield and brightfield specimens for background subtraction in Photoshop (see "Hot Pixels Removal Methods" in Chapter 6).
Grayscale: Filters
Filters can be placed in the light path to darken or lighten colors of interest. Colors that are complements (opposites) of staining colors will make the staining colors darker. Colors that are similar to the staining color will lighten stained colors (Figure 4.9).
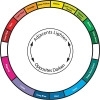
Figure 4.9 The color wheel showing color relationships.
Strong filters are required, especially when staining is weak. A reference chart is included with recommended Wratten filters, which are available from professional photography stores.
If a color camera is used for image acquisition and filters are used, ignore the resulting, strong color cast: Use the Channel Mixer dialog box (Image > Adjustments > Channel Mixer) in Photoshop to isolate the ideal grayscale image based on color that is meant to be darkened or lightened (see "Brightfield: Color to Grayscale" and "Single Color, Darkfield Images" in Chapter 7).